|
#1
April 8th, 2015, 08:17 AM
| |||
| |||
| BSC Chemistry Syllabus Pune University
I am the student of B.Sc of University of Pune and I need the B.Sc Chemistry Syllabus of it can you please provide me this? Following is the syllabus of B.Sc Chemistry of University of Pune which you are looking for Chemistry Paper - I Physical and Inorganic Chemistry Term - I Chapter 1 States of Matter 08 Chapter 2 Surface Chemistry 08 Chapter 3 Chemical Mathematics 08 Chapter4 Mole Concept, Stoichiometric and Numerical, 12 Oxidation- reduction Term - II Chapter 4 Atomic Structure 12 Chapter 5 Chemical Thermodynamics 12 Chapter 6 Chemical Bonding 12 Organic and Inorganic Chemistry Term - I Chapter1 Chemical Bonding in Organic Molecules 12 Chapter 2 Chemistry of Hydrocarbons 12 Chapter 3 Chemistry of s-block elements 12 Term - II Chapter 4 Chemistry of Functional Groups 12 Chapter 5 Stereochemistry 12 Chapter 6 Chemistry of p-block elements 12 Practical Course 1. Physical Chemistry : 7 experiments 2. Inorganic Chemistry: 7 experiments 3. Organic Chemistry : 7 experiments TERM - I Chapter 1: States of Matter (08) Introduction: States of matter and their properties. Gaseous states: Significance of ideal and kinetic gas equation (no derivation), Real gasesCompressibility factor, van der Waal’s equation of state, Isotherms of CO2, critical constants, correlation between critical constants and van der Waal’s constants. Liquid state – Properties of liquids , Comparison between gaseous and solid state – Experimental determination of vapor pressure by isoteniscopic method and viscosity by Ostwald method, liquid crystals and their applications. Chapter 2: Surface Chemistry (08) Adsorption: Types of adsorption, adsorption isotherms, Freundluich isotherm, Langmuir isotherm, adsorption of gases on solids, adsorption of solutes on solids, applications of adsorption, Catalysis : Phenomena of catalysis, types of catalysis-homogeneous and heterogeneous catalysis, gaseous reactions on solid surfaces. Colloids: Definition and classification, preparation of emulsions, gels and sols, properties of suspensoids. Aims & Objectives: Theoretical basis of adsorption phenomena is integrated. Understanding dynamic nature of surface and its applications in catalysis and in dispersed phases will lead to new area of nanoscience. Chapter 3: Chemical Mathematics (08) Functions and variables: Variables as function , variables used in chemistry Derivative: Rules of differentiation, examples on derivatives of algebraic, logarithmic and exponential functions, partial differentiation, conditions for maxima and minima, problems related to chemistry, Integration: Rules of integration (algebraic, exponential and logarithmic functions), Integration –definite and indefinite, problems related to chemistry. Graph: Plotting graphs of linear, exponential and logarithmic functions and their characteristics, sketching of s and p orbitals. Chapter 4: Mole Concept and Oxidation-reduction (12) Mole concept-Determination of mol. Weight by gram molecular volume relationship, problems based on mole concept. Methods of expressing concentrations, strength, normality, molarity, molality, %w/v, %v/v, ppm, standardization of solutions, primary & secondary standard substances, Preparation of standard solution of acids & bases, problems related to acid base titrations only. Oxidation & reduction-Definitions to related terms like oxidation, reduction, oxidizing agent, reducing agent, oxidation number, Balancing of redox reactions using oxidation number method & ion electron method, problems based one equivalent weight of oxidant & reductants. University of Pune B.Sc Chemistry Syllabus    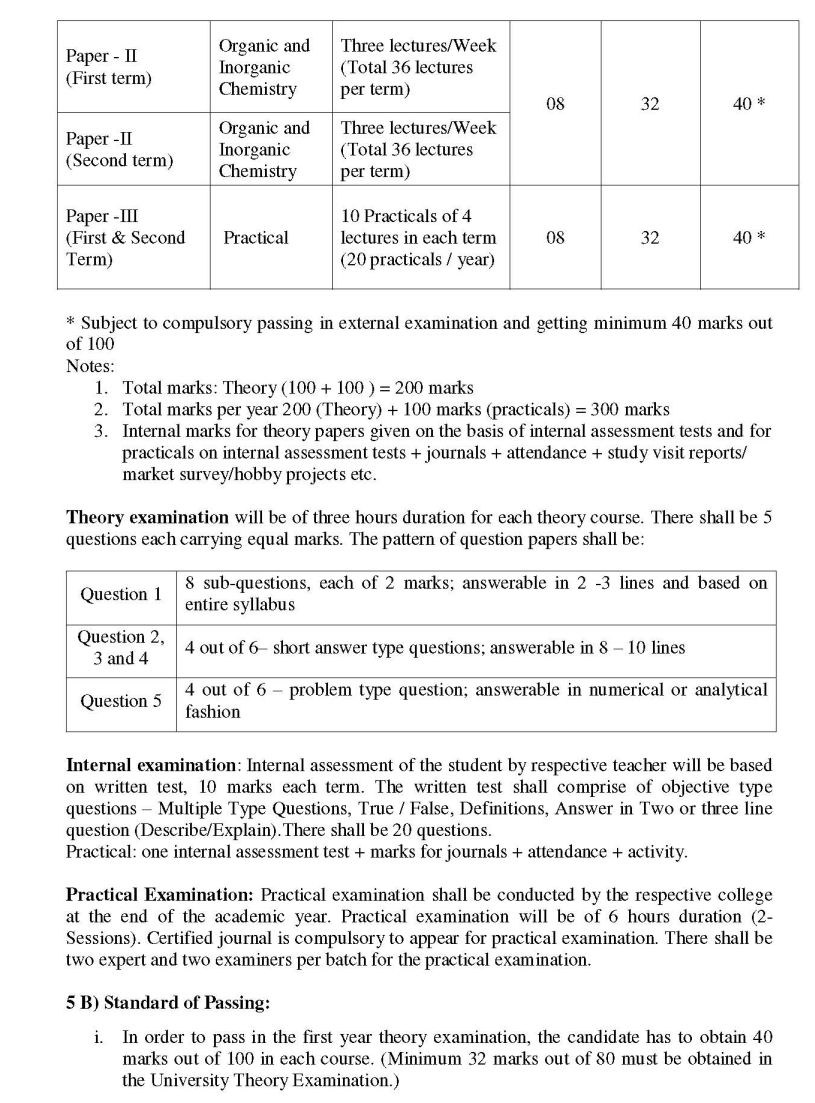  For complete syllabus here is the attachment................................. Last edited by Neelurk; February 28th, 2020 at 09:14 AM. |