|
#1
June 17th, 2016, 10:38 AM
| |||
| |||
| NIPER Hyderabad Syllabus
Will you please give here syllabus for MS (Pharma ) Pharmaceutical Analysis course offered by NIPER Hyderabad? As per your requirement I am here giving you syllabus for MS (Pharma ) Pharmaceutical Analysis course offered by NIPER Hyderabad. NIPER Hyderabad MS (Pharma ) Pharmaceutical Analysis Syllabus: I Semester PA-510 Topics in Pharmaceutical Analysis : 1. Introduction to pharmaceutical analysis and techniques: Scope and range of modern pharmaceutical analysis. Listing of various techniques, with broad discussion on their applications. 2. Material and product specifications: Definition of specifications, study of Q6 guidelines and understanding of specifications through study of pharmacopoeial monographs on drug substances and products. 3. Reference standards: Types, preparation, containers, labeling, storage and use. 4. Documentation-STPs, certificate of analysis, laboratory books: Typical documents used in a GLP laboratory including standard test protocols, COA and laboratory notebooks. 5. Introduction to method development: Method development concepts, steps involved, intricacies at each step, use of software. 6. Methods validation: Definition and methodology, discussion on each parameter with examples. 7. Calibration and qualification of equipment: Difference of definitions, calibration standards, calibration frequency, examples of calibration of pH meter, FTIR and a UV spectrophotometer. Definition of qualification process involving DQ, IQ, OQ, CQ and PQ. Brief discussion on protocol of each. 8. Bioanalysis and bioanalytical method validation: Types of body fluids, requirement of analysis, matrix effects, sample preparation, non-biological analytical samples. Acceptance criteria in comparison to non-biological samples. 9. Impurity profiling: Types of impurities in drug substances and products. Method development for impurity analysis, techniques, identification and quantitation. 10. Automation and computer-aided analysis, LIMS: The concept of auto samplers and highthroughput analysis, computer controlled instrumentation, and networked laboratory. Peculiarities of laboratory information management systems (LIMS). 11. Management of analytical laboratory: Organization of laboratories based on their types, staffing, skill development and training, budgeting and financing, purchase of costly equipment, qualities of laboratory manager and management styles. 12. Laboratory inspections: Internal inspection, external audit, concepts, preparing for inspections and audits. (MS (Pharma ) Pharmaceutical Analysis :NIPER Hyderabad) 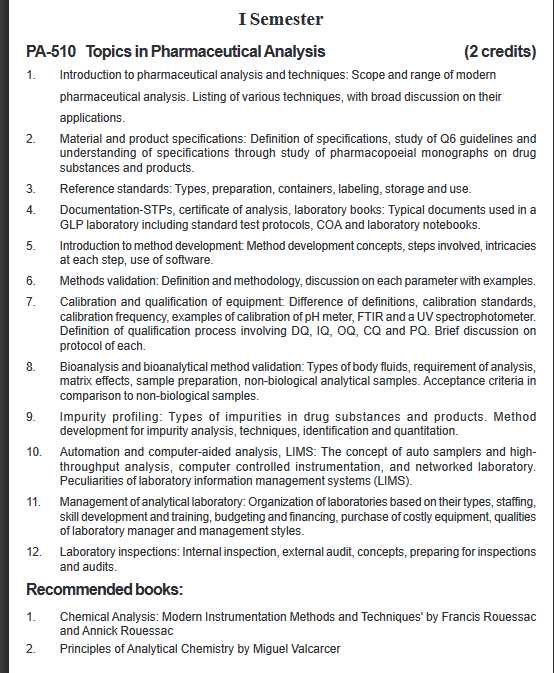     Address: National Institute of Pharmaceutical Education and Research Balanagar Hyderabad, Telangana 500037 Here is the attachment. Last edited by Neelurk; June 6th, 2020 at 09:16 AM. |