|
#1
July 5th, 2014, 12:30 PM
| |||
| |||
| WBJEE Previous Solved Question Paper
Here I am looking for the last year Chemistry solved question paper of WBJEE Exam, can you please provide me the same??? As you are looking for the last year Chemistry solved question paper of WBJEE Exam, so here I am sharing the same with you In diborane, the number of electrons that account for bonding in the bridges is (A) Six (B) Two (C) Eight (D) Four Ans : (D) A van der Waals gas may behave ideally when (A) The volume is very low (B) The temperature is very high (C) The pressure is very low (D) The temperature, pressure and volume all are very high Ans : (C) 2-Methylpropane on monochlorination under photochemical condition give (A) 2-Chloro-2-methylpropane as major product (B) (1:1) Mixture of 1-chloro-2-methylpropane and 2-chloro-2-methylpropane (C) 1-Chloro-2-methylpropane as a major product (D) (1:9) Mixture of 1-chloro-2-methylpropane and 2-chloro-2-methylpropane Ans : (C) Chlorine gas reacts with red hot calcium oxide to give (A) Bleaching powder and di hlorine monoxide (B) Bleaching powder and water (C) Calcium chloride and chlorine dioxide (D) Calcium chloride and oxygen Ans : (D)   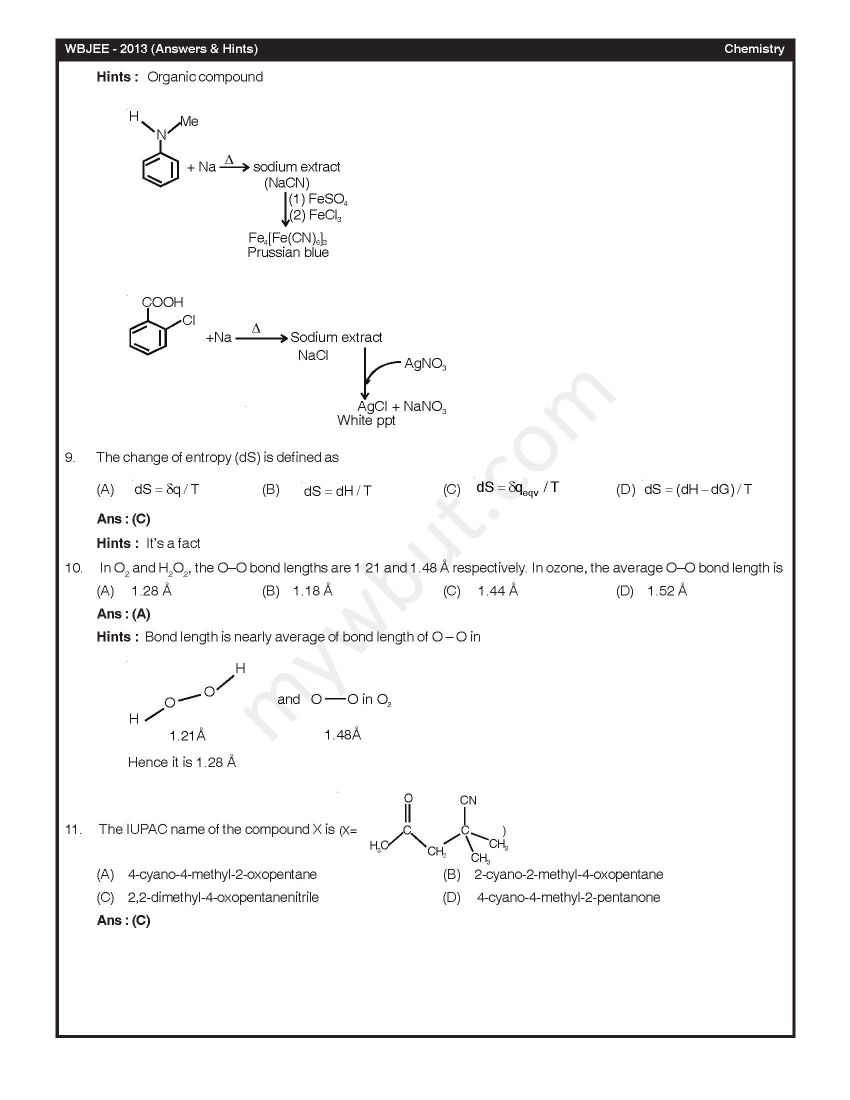  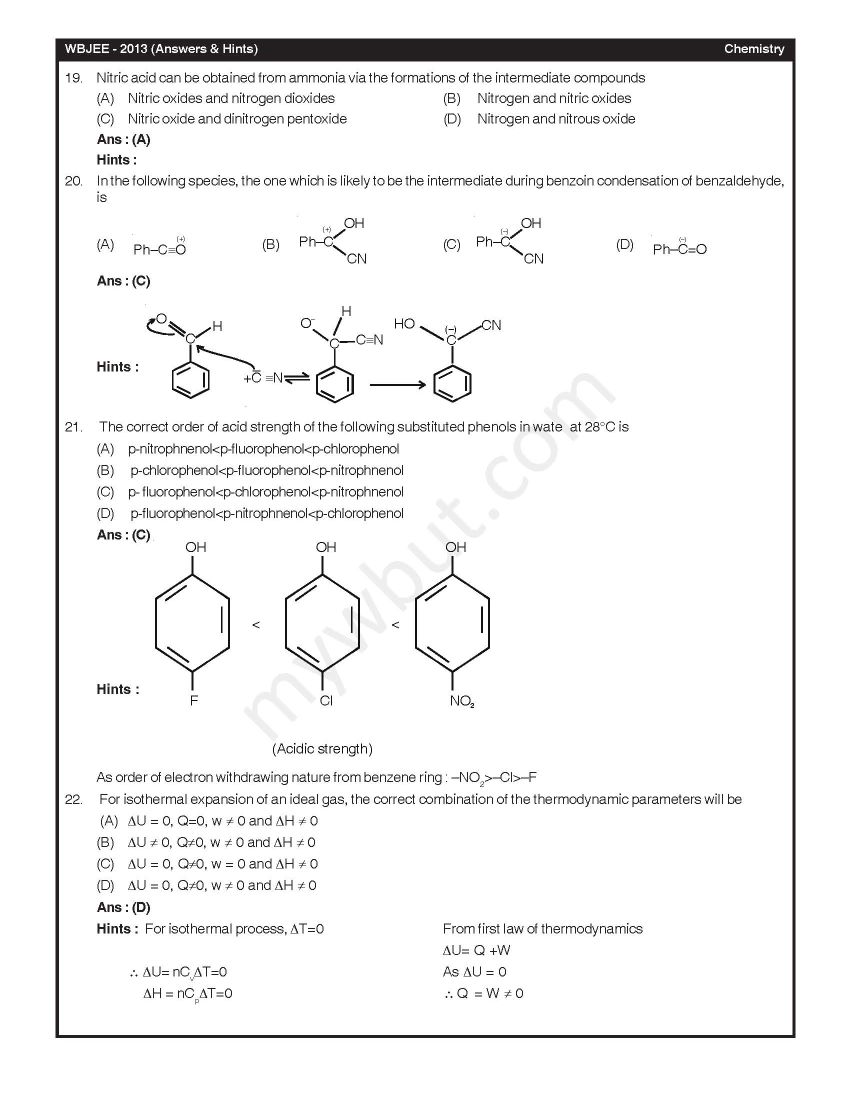 Rest of the Questions are attached in below file which is free of cost Last edited by Neelurk; March 25th, 2020 at 02:59 PM. |