|
#2
December 13th, 2017, 03:43 PM
| |||
| |||
| Re: Zinc and HCL Balanced Equation
The reaction amongst zinc and hydrochloric corrosive is Zn + HCl = H2 + ZnCl2. It is a solitary substitution response where zinc metal dislodges the hydrogen to frame hydrogen gas and zinc chloride, a salt. Zinc responds rapidly with the corrosive to frame rises of hydrogen. Is zinc and hydrochloric acid a redox response? It is genuinely clear that zinc metal responds with fluid hydrochloric corrosive! The air pockets are hydrogen gas. ... Seen along these lines, the net response is by all accounts a charge exchange amongst zinc and hydrogen molecules. (There is no net change experienced by the chloride particle.) Is zinc and hydrochloric acid an exothermic response? Synthetic responses can likewise be exothermic or endothermic, radiating or taking in vitality as warmth. The substance response between Zinc metal and Hydrochloric corrosive appeared above is Obviously exothermic in light of the fact that it emits warm. What is the balance equation for zinc and hydrochloric corrosive? Keeping in mind the end goal to adjust Zn + HCl = ZnCl2 + H2 you'll have to ensure the quantity of Zn, H and Cl iotas are the same on the two sides of the substance condition. You do that by changing the coefficients (the numbers before particles or mixes). Oxidation-Reduction (Redox) Reactions 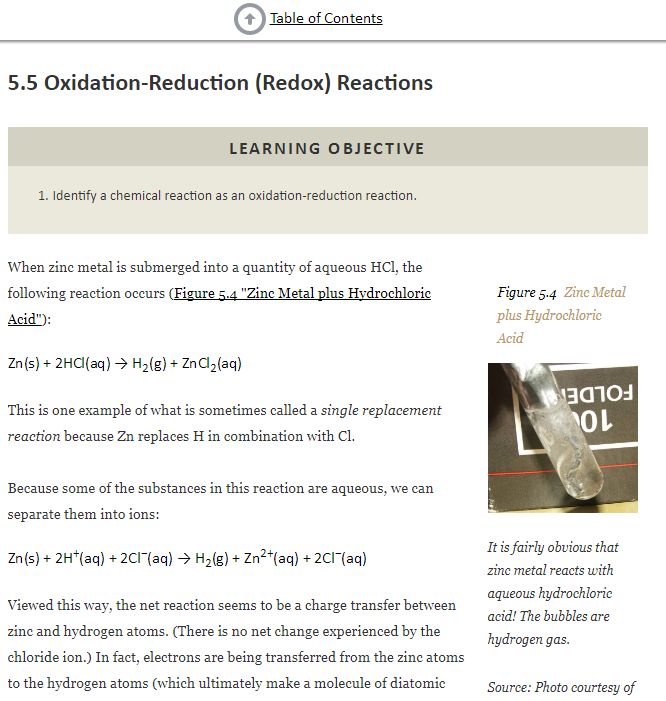  |