|
#2
January 25th, 2018, 11:58 AM
| |||
| |||
| Re: General Organic Chemistry Notes IIT JEE
I am providing you the notes of General Organic Chemistry of IIT Joint Entrance Exam IIT JEE Main Exam General Organic Chemistry Notes General Organic Chemistry: Bond Fission: a) Homolytic fission: Each atom separates with one electron, leading to the formation of highly reactive entities called radicals, owing their reactivity to their unpaired electron. b) Heterolytic fission: One atom holds on to electrons, leaving none for the other, the result in the above case being a negative and positive ion, respectively, the result being the formation of an ion pair. Reactions involving radicals tend to occur in the gas phase and in solution in non-polar solvents, and to be catalyzed by light and by the addition of other radicals. Reactions involving ionic intermediates take place more readily in solution in polar solvents, because of the greater ease of separation of charges therein and very often because of the stabilization of the resultant ion pairs through solvation.Homolytic and Heterolytic Bond Fision Electronic Displacement in Covalent Bonds The following four types of electronic effects operates in covalent bonds a) Inductive effect b) Mesomeric and Resonance effect c) Electromeric effects d) Hyperconjugation Inductive Effect: a) Negative inductive Effect: (–I Effect):This is due to electron - attracting groups (X); it develops positive charge on the chain and is said to exert a negative inductive denoted by (– I) 1) It decreases as one goes away from group X (electron attracting): X-C1-C2-C3 C1(d+) > C2(dd+) > C3(ddd+) and after third carbon charge is negligible For complete notes jeer is the attachment IIT JEE Main Exam General Organic Chemistry Notes  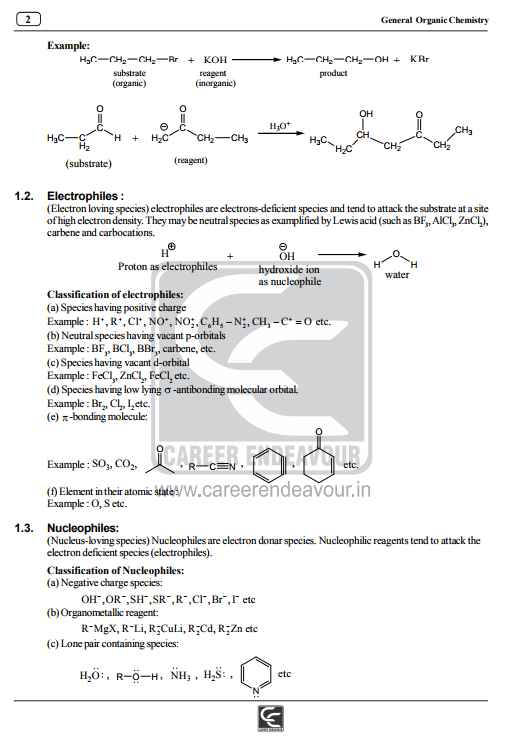 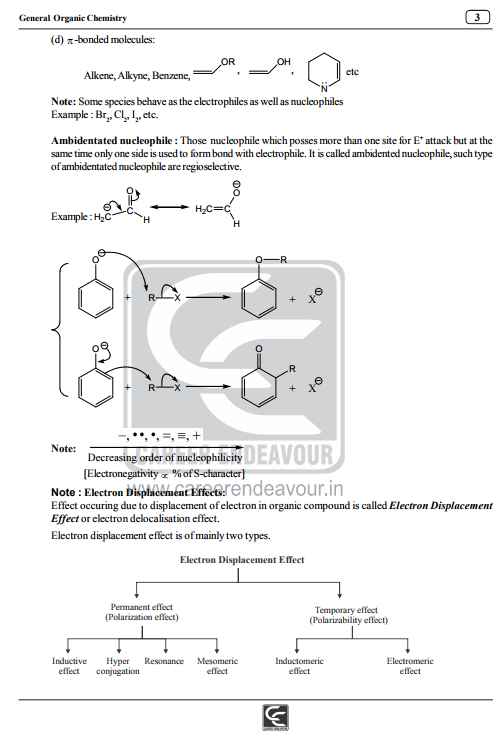 |